Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (31): 5065-5071.doi: 10.3969/j.issn.2095-4344.1442
Previous Articles Next Articles
Mechanism of long non-coding RNA in exercise-mediated bone autophagy
Yang Kang1, Chen Xianghe1, Zhao Renqing1, Yu Huilin1, Zhang Xianliang2, Bu Wenqian1
- (1School of Physical Education, Yangzhou University, Yangzhou 225127, Jiangsu Province, China; 2School of Physical Education, Shandong University, Jinan 250100, Shandong Province, China)
-
Received:2019-06-03Online:2019-11-08Published:2019-11-08 -
Contact:Chen Xianghe, PhD, School of Physical Education, Yangzhou University, Yangzhou 225127, Jiangsu Province, China -
About author:Yang Kang, Master candidate, School of Physical Education, Yangzhou University, Yangzhou 225127, Jiangsu Province, China -
Supported by:the Graduate Research and Innovation Project of Yangzhou University, No. XKYCX18_055 (to YK); the General Project of Natural Science Foundation of Jiangsu Provincial Universities, No. 17KJB180017 (to CXH); the Social Science Research Project of Shandong Province, No. 17DTYJ01 (to ZXL)
CLC Number:
Cite this article
Yang Kang, Chen Xianghe, Zhao Renqing, Yu Huilin, Zhang Xianliang, Bu Wenqian . Mechanism of long non-coding RNA in exercise-mediated bone autophagy [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(31): 5065-5071.
share this article
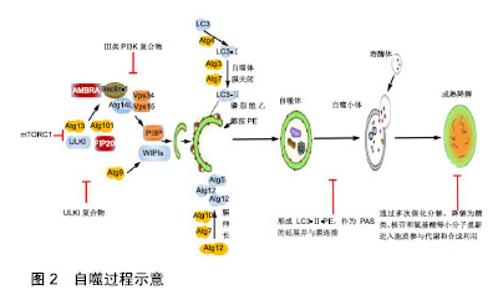
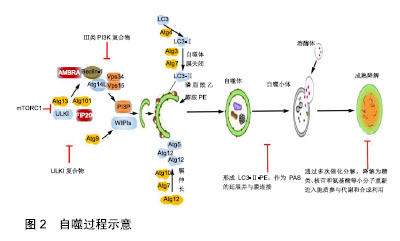
2.1 自噬发生机制 骨自噬分为微自噬(microautophagy)、分子伴侣介导的自噬和巨自噬(macroautophagy)3种方式。自噬发生过程包括自噬诱导、自噬体形成和成熟降解3个阶段,见图2。细胞自噬在正常生理条件下会保持基础水平,而在遇到如运动刺激、饥饿、氧化应激等情况时,将开始自噬诱导。以上条件下,其上游原本活跃的mTOR被抑制,mTORC1活性降低,导致ULK1/2、mATG13的去磷酸化并组成mATG13-ULK1/2复合物,活化ULK1/2,活化的后者又磷酸化FIP200,进而启动自噬[8]。有研究表明,自噬启动时,不同于mTOR通过ULK1Ser757位点抑制ULK1的方式,AMPK可磷酸化ULK1Ser317和Ser777位点,进而激活ULK1[9]。自噬诱导后,自噬相关基因14、Beclin1、Vps15和mVps34组成复合体启动成核反应,将自噬相关基因21、自噬相关基因24结合到膜上组成前自噬体(pre-autophagosomal structure,PAS)[10]。此处需要2种泛素化系统:自噬相关基因12-自噬相关基因5和微管相关蛋白轻链3Ⅱ-磷脂酰乙醇胺参与[11]。形成的自噬体经微管转运到溶酶体,外膜与溶酶体相融合,由水解酶完成整个自噬小体的成熟降解。此阶段中,自噬体内溶胶环境的酸化是自噬体与囊泡融合的必要步骤,且有Lamp2、小GTP蛋白Rab7和紫外线放射抗性相关蛋白等因子参与[12]。通过多次催化分解,降解为糖类、核苷和氨基酸等小分子重新进入胞质参与代谢和合成利用。 2.2 骨自噬 2.2.1 骨髓间充质干细胞自噬 骨髓间充质干细胞具有自我更新和多向分化的特性和修复重建受损骨组织的功能,是治疗骨疾病的首选种子细胞,其自噬水平的变化直接影响成骨分化功能的发挥。有学者报道,骨自噬在提高骨髓间充质干细胞氧化应激存活率的同时,还 "

能保持骨髓间充质干细胞活性的稳定[13]。研究显示,5%的低氧条件可通过抑制丝裂原活化蛋白激酶、激活PI3K/AKT/mTOR等通路,联合成骨分化诱导液促进骨髓间充质干细胞向成骨分化[14]。在缺氧模型中,白细胞介素8能通过Akt/STAT3通路提高人骨髓间充质干细胞自噬和增殖能力,降低缺氧缺血对人骨髓间充质干细胞造成的损伤[15]。与之相反,通过Western blot分别检测年轻小鼠和衰老小鼠微管相关蛋白轻链3Ⅱ、Beclin 1、p62蛋白表达发现,与年轻组小鼠相比,老年组小鼠骨髓间充质干细胞凋亡增加,微管相关蛋白轻链3Ⅱ、Beclin 1表达下降,p62蛋白表达水平升高,自噬信号通路相关蛋白p-Akt、Akt、p-mTOR、mTOR表达升高,骨量减少[16]。这表明在衰老和细胞器受损条件下,老年小鼠骨髓间充质干细胞自噬活性下降,凋亡增加,抑制骨髓间充质干细胞成骨分化。有学者研究发现,氧化应激对骨髓间充质干细胞自噬具有双重作用,即在诱导其凋亡的同时亦诱导保护性自噬作用,抑制骨髓间充质干细胞自噬可加速其凋亡,增强其自噬能提高其在2型糖尿病移植治疗中的存活率[17]。此外,mTOR能提高自噬活性较低的骨髓间充质干细胞自噬活性,减弱其向成骨细胞分化的潜力[18]。以上均提示自噬可能在骨髓间充质干细胞向成骨细胞分化的过程中扮演重要角色。 2.2.2 成骨细胞自噬 成骨细胞由骨髓间充质干细胞分化而来,通过分泌胶原、基质和酶类启动并主导骨形成过程。研究证明,自噬能正向调控成骨细胞分化和成熟。如糖尿病高糖环境过度产生活性氧,加速成骨MC3T3-E1细胞中的蛋白氧化和自噬,而自噬缺陷会导致线粒体形态学缺陷,抑制成骨细胞在高糖环境中的分化[19]。表明自噬是维持成骨细胞在高糖环境下活性和功能的重要机制。在观察颅骨成骨分化时,发现自噬流增加显著,这提示在自噬参与颅骨早期成骨分化。此外,成骨细胞中核因子kB信号通路激活需要其与巨噬细胞接触,且此过程受p62抑制,即当成骨细胞内p62缺陷将导致骨丢失,并影响成骨细胞中CCL4表达及造血祖细胞趋化性,最终引发造血干细胞流失[20]。而骨重建是一个连续的生理过程,需骨髓间充质干细胞持续向成骨细胞分化。而此过程需增强线粒体呼吸以保持能量源源不断供应,这必然导致内源性活性氧堆积。FOXO3作为骨髓间充质干细胞向成骨细胞分化的氧化还原平衡器,一旦下降,将导致活化氧水平升高,进而使骨髓间充质干细胞向成骨细胞分化减慢甚至停滞。与之相同的是,敲除自噬相关基因7抑制自噬后,骨髓间充质干细胞调控活化氧能力下降,成骨细胞分化也随之降低。这提示FOXO3能通过诱导自噬抑制活化氧,进而加速骨髓间充质干细胞向成骨细胞分化。后续研究发现,人骨髓间充质干细胞通过AMPK途径前期抑制mTOR,后期激活Akt/mTOR轴介导自噬促骨髓间充质干细胞向成骨细胞[21]。 2.2.3 破骨细胞自噬 破骨细胞是巨大多核细胞,由RANKL与成骨细胞产生的巨噬细胞集落刺激因子激活融合而成,主导骨吸收[22]。研究表明,自噬能影响破骨细胞活性及功能的发挥。p62这一自噬特异性调控蛋白,是联结RANKL诱导的自噬和破骨细胞生成的桥梁,敲除p62不仅抑制破骨细胞中RANKL诱导的自噬活化,还抑制破骨细胞生成[23]。p62在佩吉特骨病中突变,引发破骨细胞体积增大,细胞活性升高。而p62是自噬调节关键mTOR通路的重要参与因子,这提示破骨细胞上的p62突变可影响破骨细胞自噬,以调节破骨细胞分化[24]。在人类类风湿关节炎中,肿瘤坏死因子α可激活自噬使破骨细胞生成增加,而以药物抑制自噬可抑制破骨细胞分化,敲除肿瘤坏死因子α转基因小鼠骨髓细胞中自噬相关基因7后发现,破骨细胞数目减少[25]。表明肿瘤坏死因子α在诱导自噬过程中,与破骨细胞呈正相关。肿瘤坏死因子受体相关因子3限制RANKL诱导破骨细胞生成,而肿瘤坏死因子受体相关因子3自噬性降解能加速破骨细胞生成[26]。由多杀巴氏杆菌毒素启动的mTOR信号活化在破骨细胞形成过程中起重要作用,可能通过下调程序性细胞死亡蛋白4表达,程序性细胞死亡蛋白4是破骨细胞特异性基因转录因子c-Jun的抑制物,从而激活c-Jun,启动破骨细胞特异性基因的转录[27]。而缺氧应激通过HIF-1/BNIP3激活自噬,进而促进破骨细胞分化[28]。 2.3 长链非编码RNA调控骨自噬的作用机制 转录生成非编码RNA的序列约占人类基因组的绝大部分,其中包括housekeeping非编码RNA、小分子非编码RNA、中等长度非编码RNA和长链非编码RNA。长链非编码RNA在细胞增殖分化、个体发育、信号转导、干细胞维持、代谢等重要生命活动中发挥关键调控作用。其在表观遗传水平、转录水平前后水平等方面具有控制基因表达的作用。此外,肿瘤、糖尿病、精神疾病和神经退行性疾病的发生都与长链非编码RNA的异常表达有关[29]。研究发现,长链非编码RNA在骨细胞的生长发育与分化过程中意义重大,以顺式和反式作用参与并作用于骨细胞分裂、剂量补偿和细胞分化等过 程[30]。长链非编码RNA的一级结构——核苷酸排列顺序(碱基互补配对)是长链非编码RNA调节靶基因及其上下游基因转录翻译的基础。其作为竞争性内源RNA占据靶基因的结合位点,抑制下游的小分子RNA或mRNA与靶基因结合(也有学者将此行为称为靶基因模仿)[31]。这种负向调控下游小分子RNA的方式可能是长链非编码RNA调控细胞自噬的重要途径,如与肿瘤发生相关的长链非编码RNA生长阻滞特异转录物5被证明能结合糖皮质激素的DNA结合域,竞争糖皮质激素反应元件目的基因并调节其表达[32]。这种竞争结合靶基因位点的方式在肌肉分化中也有所体现,Linc-MD1通过碱基互补配对的方式与miR-133和miR-135结合,竞争性抑制二者与靶基因的结合,进而影响肌细胞自 噬[33]。 通过查阅整理大量文献发现,有关长链非编码RNA调控细胞自噬的研究多集中在肿瘤细胞、心肌细胞、脑细胞和心肌细胞等,且具体长链非编码RNA如何调控上述细胞机制阐述尚不清晰,见表1。 比较正常组织和膀胱癌组织时发现,后者中长链非编码RNA MEG3表达显著降低、微管相关蛋白轻链3Ⅱ显著升高。而其过表达则导致自噬水平降低,这表明长链非编码RNA MEG3与微管相关蛋白轻链3Ⅱ呈负相关。长链非编码RNA MEG3可与自噬相关基因17相互作用以抑制自噬,而自噬蛋白p53作为调控长链非编码 "

| [1]Liu Y, Wang N, Zhang S, et al.Autophagy protects bone marrow mesenchymal stem cells from palmitate-induced apoptosis through the ROS-JNK/p38 MAPK signaling pathways. Mol Med Rep. 2018;18(2):1485-1494.[2]Camuzard O, Santucci-Darmanin S, Breuil V, et al. Sex-specific autophagy modulation in osteoblastic lineage: a critical function to counteract bone loss in female. Oncotarget. 2016;7(41): 66416-66428.[3]Xu F, Li X, Yan L, et al. Autophagy Promotes the Repair of Radiation-Induced DNA Damage in Bone Marrow Hematopoietic Cells via Enhanced STAT3 Signaling. Radiat Res. 2017;187(3):382-396.[4]Wang M, Han D, Yuan Z, et al.Long non-coding RNA H19 confers 5-Fu resistance in colorectal cancer by promoting SIRT1-mediated autophagy. Cell Death Disease. 2018;9(12): 1149.[5]Wang YG, Hu YN, Sun CX, et al.Down-regulation of Risa improves insulin sensitivity by enhancing autophagy. FASEB J. 2016;30(9):3133-3145.[6]Yu X, Pang L, Yang T, et al.lncRNA LINC01296 regulates the proliferation, metastasis and cell cycle of osteosarcoma through cyclin D1. Oncol Rep. 2018;40(5):2507-2514.[7]郭健民.运动对骨质疏松小鼠骨长链非编码RNA表达的影响[D].上海:上海体育学院,2017.[8]Abrahamsen H, Stenmark H, Platta HW.Ubiquitination and phosphorylation of Beclin 1 and its binding partners: Tuning class III phosphatidylinositol 3-kinase activity and tumor suppression. FEBS Lett. 2012;586(11):1584-1591.[9]Kim J, Kundu M, Viollet B, et al.AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol. 2011;13(2):132-141.[10]Feng H, Zhao X, Guo Q, et al. Autophagy resists EMT process to maintain retinal pigment epithelium homeostasis. Int J Biol Sci. 2019;15(3):507-521.[11]Zhang W, Chen K, Guo Y, et al.Involvement of PRRSV NSP3 and NSP5 in the autophagy process. Virol J. 2019; 16(1):13. [12]Yamamoto F, Taniguchi K, Mamada N, et al.TFEB-mediated Enhancement of the Autophagy-lysosomal Pathway Dually Modulates the Process of Amyloid β-Protein Generation in Neurons. Neuroscience. 2019;40(2):11-22.[13]Liu Z, Li T, Zhu F, et al.Regulatory roles of miR-22/Redd1-mediated mitochondrial ROS and cellular autophagy in ionizing radiation-induced BMSC injury. Cell Death Dis. 2019;10(3):227.[14]Dong Y, Wu Y, Zhao GL, et al.Inhibition of autophagy by 3-MA promotes hypoxia-induced apoptosis in human colorectal cancer cells. Eur Rev Med Pharmacol Sci. 2019;23(3): 1047-1054.[15]Shologu N, Scully M, Laffey JG, et al.Human mesenchymal stem cell secretome from bone marrow or adipose-derived tissue sources for treatment of hypoxia-induced pulmonary epithelial injury. Int J Mol Sci. 2018;19(10):E2996.[16]Akahane M, Shimizu T, Inagaki Y, et al.Implantation of Bone Marrow Stromal Cell Sheets Derived from Old Donors Supports Bone Tissue Formation. Tissue Eng Regen Med. 2017;15(1):89-100.[17]Wang H, Wang Z, Tang Q. Reduced expression of microRNA-199a-3p is associated with vascular endothelial cell injury induced by type 2 diabetes mellitus. Exp Ther Med. 2018;16(4):3639-3645.[18]Ka M, Smith AL, Kim WY. MTOR controls genesis and autophagy of GABAergic interneurons during brain development. Autophagy. 2017;13(8):1348-1363.[19]Bartolomé A, López-Herradón A, Portal-Nú, et al.Autophagy impairment aggravates the inhibitory effects of high glucose on osteoblast viability and function. Biochem J. 2013;455(3): 329-337.[20]Chang KH, Sengupta A, Nayak RC, et al. p62 is required for stem cell/progenitor retention through inhibition of IKK/NF- B/Ccl4 signaling at the bone marrow macrophage-osteoblast niche. Cell Rep. 2014;9(6):2084-2097.[21]Gómez-Puerto MC, Verhagen LP, Braat AK, et al. Activation of autophagy by FOXO3 regulates redox homeostasis during osteogenic differentiation. Autophagy.2016;12(10): 1804-1816.[22]Pierrefite-Carle V,Santucci-Darmanin S, BreuilV, et al. Autophagy in bone: Self-eating to stay in balance. Ageing Res Rev. 2015; 24 (2): 206-217.[23]Zhu YB, Jia GL, Lu JH, et al. The relationship between autophagy activation in spinal cord and type 2 diabetic neuropathic pain in rats. Acta Physiologica Sinica. 2018; 34(4):318-323.[24]Helfrich MH, Hocking LJ. Genetics and aetiology of Pagetic disorders of bone. Arch Biochem Biophys. 2008;473(2): 172-182.[25]Lin NY, Beyer C, Giessl A, et al. Autophagy regulates TNF-mediated joint destruction in experimental arthritis. Ann Rheum Dis. 2013;72 (5):761-768.[26]Xiu Y, Xu H, Zhao C, et al. Chloroquine reduced osteoclast genesis in murine osteoporosis by preventingTRAF3degradation.Clin Invest. 2014;124(1):297.[27]Kloos B, Chakraborty S, Lindner SG, et al. Pasteurellamultocida toxin-induced osteoclast genesis requires mTOR activation.Cell Commun Signal. 2015;13(1): 40.[28]Zhao Y, Chen G, Zhang W, et al. Autophagy regulates hypoxiainduced Osteoclast genesis through the HIF-1/BNIP3 signaling pathway. J Cell Physiol. 2012;227(2): 639-648.[29]Yu C, Li L, Xie F, et al. LncRNA TUG1 sponges miR-204-5p to promote osteoblast differentiation through upregulating Runx2 in aortic valve calcification. Cardiovasc Res. 2018; 114(1):703-711.[30]Ma l, Bajic VB, Zhang Z. On the classification of long noncoding RNAs. RNA Biol. 2013;10(6):925-933.[31]Wu DM, Wang S, Wen X, et al. LncRNA SNHG15 acts as a ceRNA to regulate YAP1-Hippo signaling pathway by sponging miR-200a-3p in papillary thyroid carcinoma. Cell Death Dis. 2018;9(10):947.[32]Novikova IV, Hennelly SP, Sanbonmatsu KY. Sizing up long non-coding RNAs: Do LncRNAs have secondary and tertiary structure. Bioarchitecture. 2012;2(6):189-199.[33]Legnini I, Morlando M, Mangiavacchi A, et al. A Feedforward Regulatory Loop between HuR and the Long Noncoding RNA linc-MD1 Controls Early Phases of Myogenesis. Mol Cell. 2014;53(3):506-514.[34]Pawar K, Hanisch C, Palma SE, et al. DownRegulated lncRNA MEG3 eliminates mycobacteria in Macrophages via autophagy. Sci Rep. 2016;13(6):19416.[35]Zhuo CJ, Jiang RH, Lin XD, et al. LncRNA H19 inhibits autophagy by epigenetically silencing of DIRAS3 in diabetic cardiomyopathy. Oncotarget. 2016;8(1):1429-1437.[36]Li S, Pan X, Yang S, et al. LncRNA MALAT1 promotes oxidized low-density lipoprotein-induced autophagy in HUVECs by inhibiting the PI3K/AKT pathway. J Cell Biochem. 2019;120(3):4092-4101.[37]Yuan P, Cao WB, Zang QL, et al. The HIF-2α-MALAT1- miR-216b axis regulates multidrug resistance of hepatocellular carcinoma cells via modulating autophagy. Biochem Biophys Res Comm. 2016;478(3):1067-1073.[38]Wang Y, Hu Y, Sun C, et al. Down-regulation of Risa improves insulin sensitivity by enhancing autophagy. FASEB J. 2016; 30(9):3133-3145.[39]阮坚.不同强度耐力运动对老龄大鼠骨骼肌自噬的影响[D].长沙:湖南师范大学,2018.[40]Xu J, Jiao K, Liu X, et al. Omi/HtrA2 Participates in age-related autophagic deficiency in rat liver. Aging Dis. 2018;9(6):1031-1042.[41]陈祥和.不同方式运动对Ⅱ型糖尿病小鼠骨代谢的影响及分子机制研究[D].上海:华东师范大学,2016.[42]Zhang B, Zhang Y, La Cour KH, et al. Mitochondrial aldehyde dehydrogenase obliterates endoplasmic reticulum stress-induced cardiac contractile dysfunction via correction of autophagy. Biochim Biophys Acta. 2013;1832(4):574-584.[43]Wu H, Wu Z, Li P, et al. Bone size and quality regulation: concerted actions of mTOR in mesenchymal stromal cells and osteoclasts. Stem Cell Reports. 2017;8(6):1600-1616.[44]孙自强. AKT/mTOR/ULK1介导的自噬在骨保护素调控破骨细胞骨吸收活性中的作用机制[D].扬州:扬州大学,2017.[45]Liu YM, Ma JH, Zeng QL, et al. MiR-19a Affects Hepatocyte Autophagy via Regulating LncRNA NBR2 and AMPK/PPARα in D-GalN/Lipopoly saccharide-Stimulated Hepatocytes. J Cell Biochem. 2018;119(1):358-365. [46]Meng X, Guo J, Fang W, et al. Liver microRNA-291b-3p promotes hepatic lipogenesis through negative regulation of adenosine 5'-monophosphate (AMP)-activated protein kinase. J Biol Chem. 2016;291(20):10625-10634.[47]An T, He ZC, Zhang XQ, et al. Baduanjin exerts anti-diabetic and anti-depression effects by regulating the expression of mRNA, lncRNA, and circRNA. Chin Med. 2019;14(1):3.[48]Shang JL, Cheng Q, Duan SJ, et al. Cognitive improvement following ischemia/reperfusion injury induced by voluntary running-wheel exercise is associated with Lnc MALAT1-mediated apoptosis inhibition. Int J Mol Med. 2018; 41(5):2715-2723. [49]Shuping P, Lihua C, Shiwei H,et al. An Overview of Long Noncoding RNAs Involved in Bone Regeneration from Mesenchymal Stem Cells. Stem Cells Int. 2018;2018:1-11. |
| [1] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [2] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [3] | Wang Mengting, Gu Yanping, Ren Wenbo, Qin Qian, Bai Bingyi, Liao Yuanpeng. Research hotspots of blood flow restriction training for dyskinesia based on visualization analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1264-1269. |
| [4] | Wang Yongsheng, Wu Yang, Li Yanchun. Effect of acute high-intensity exercise on appetite hormones in adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1305-1312. |
| [5] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [6] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [12] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [13] | Xie Yang, Zhang Shujiang, Liu Menglan, Luo Ying, Yang Yang, Li Zuoxiao. Mechanism by which rapamycin protects spinal cord neurons in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 695-700. |
| [14] | Jiang Xin, Qiao Liangwei, Sun Dong, Li Ming, Fang Jun, Qu Qingshan. Expression of long chain non-coding RNA PGM5-AS1 in serum of renal transplant patients and its regulation of human glomerular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 741-745. |
| [15] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||